Amoxycillin And Potassium Clavulanate Injection IP
Amoxycillin And Potassium Clavulanate Injection IP Specification
- Brand Name
- Not specified (generic product label)
- Indication
- Treatment of respiratory tract, urinary tract, skin, and other bacterial infections
- Pacakaging (Quantity Per Box)
- Usually 1 vial per box
- Salt Composition
- Amoxycillin Sodium and Potassium Clavulanate
- Life Span
- 24 months from date of manufacture
- Dosage Form
- Injection IP
- Packaging Type
- Glass vial with carton
- Drug Type
- Prescription (Rx)
- Ingredients
- Amoxycillin Sodium and Potassium Clavulanate
- Physical Form
- Powder
- Function
- Antibiotic, fights bacterial infections
- Recommended For
- Adults and Children with Susceptible Bacterial Infections
- Dosage
- As directed by physician
- Dosage Guidelines
- Intravenous injection or infusion; reconstitute before use
- Suitable For
- Suitable for both adults and children
- Quantity
- In single vial packs; vial sells individually
- Storage Instructions
- Store below 25C, protected from moisture and light
- Appearance
- White to off-white sterile powder in vial
- Manufacturing Standard
- WHO-GMP Certified Facility
- Route of Administration
- Intravenous (IV)
- Contraindications
- Hypersensitivity to penicillins, severe renal impairment
- Major Drug Interactions
- Should not be used with allopurinol, probenecid, methotrexate without medical supervision
- Regulatory Approval
- Manufactured under Indian Pharmacopeia (IP) standards
- Strength
- Typically available as 1.2g (1000mg Amoxycillin + 200mg Clavulanate) per vial
- Side Effects
- Possible nausea, rash, diarrhea, allergic reactions
- Shelf Life After Reconstitution
- Use immediately; discard unused portion
- Reconstitution Instructions
- Dissolve in sterile water for injection as per label directions before administration
- Marketed By
- Details as per carton label
Amoxycillin And Potassium Clavulanate Injection IP Trade Information
- Minimum Order Quantity
- 10 Pieces
- Payment Terms
- Cash in Advance (CID)
- Supply Ability
- 100 Pieces Per Month
- Delivery Time
- 5-7 Days
- Main Domestic Market
- All India
About Amoxycillin And Potassium Clavulanate Injection IP
Outstanding Advantages and Targeted Application
Amoxycillin And Potassium Clavulanate Injection IP stands out for its rapid action and WHO-GMP compliant manufacture. It is suitable for intravenous use, addressing bacterial infections across the respiratory tract, urinary tract, and skin. The primary competitive advantage lies in its broad-spectrum efficacy and precise dosing, making it ideal for hospitals and clinics. Proper reconstitution and administration by professionals ensure maximum therapeutic benefits for both adults and pediatric patients.
Swift Delivery, Trusted Certification, and Sample Availability
Each vial comes securely packaged in a glass container with carton, ensuring safety during express shipping. Orders are delivered promptly throughout India with reliable logistics partners. The product is manufactured in a certified facility following Indian Pharmacopeia standards. For those evaluating supplies, sample vials are available on request to facilitate quality checks before wholesale procurement.
FAQ's of Amoxycillin And Potassium Clavulanate Injection IP:
Q: How should Amoxycillin And Potassium Clavulanate Injection IP be administered?
A: This injection is meant for intravenous use only, either as a direct injection or infusion. It must be reconstituted with sterile water for injection according to the label instructions before administration by a healthcare professional.Q: What are the major benefits of using this antibiotic?
A: The main benefits include its powerful broad-spectrum antibacterial action, rapid onset in treating respiratory, urinary, and skin infections, and its suitability for both adults and children.Q: When should this injection not be used?
A: This product should not be used in patients with hypersensitivity to penicillins, those with severe renal impairment, or those taking medications like allopurinol, probenecid, or methotrexate without medical supervision.Q: Where is the product manufactured and what certifications does it have?
A: The injection is produced in India, adhering to WHO-GMP standards and the Indian Pharmacopeia, assuring quality and safety in every batch.Q: What should be done with any unused solution after reconstitution?
A: Any unused portion of the reconstituted solution should be discarded immediately, as its efficacy and sterility cannot be guaranteed after reconstitution.Q: How is the product packaged and delivered?
A: Each vial is packed in an individual glass vial and carton. Orders are dispatched via express shipping and promptly delivered throughout India.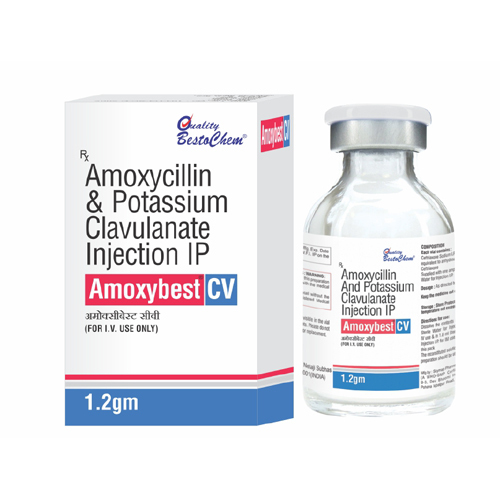

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharmaceutical Injection Category
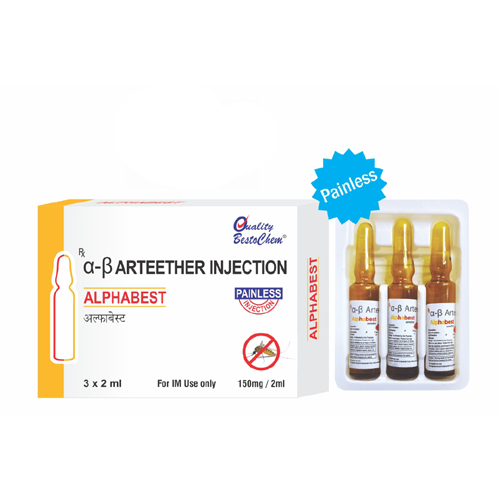
3 x 2ml A-B Arteether Injection
Minimum Order Quantity : 10 Pieces
Dosage : 150 mg (as per doctors prescription), once daily for 3 consecutive days
Indication : Cerebral malaria, severe/complicated malaria
Pacakaging (Quantity Per Box) : 3 ampoules per box
Quantity : 3 ampoules x 2ml
Brand Name : AB Arteether Injection
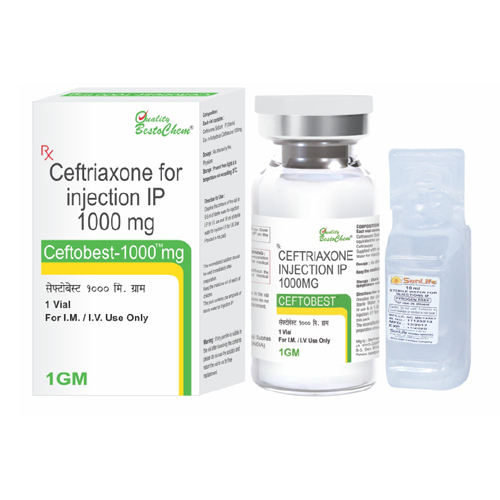
1000mg Ceftriaxone For Injection IP
Minimum Order Quantity : 10 Pieces
Dosage : As directed by the physician. Typically 1g (1000mg) once or twice daily, intravenously or intramuscularly.
Indication : Management of various susceptible bacterial infections including respiratory tract, urinary tract, skin, bone, joint, abdominal and septicemia
Pacakaging (Quantity Per Box) : Usually 1 vial per box
Quantity : 1000mg (1g)
Brand Name : As per manufacturer
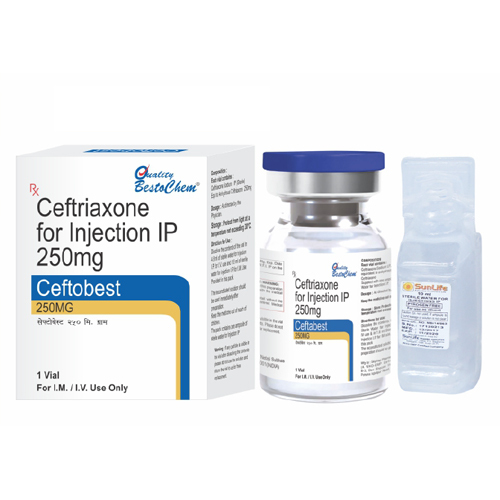
250mg Ceftriaxone For Injection IP
Minimum Order Quantity : 10 Pieces
Dosage : As directed by physician
Indication : Respiratory tract, urinary tract, skin, bone, joint, intraabdominal, and septicemia infections
Pacakaging (Quantity Per Box) : 10 vials per box
Quantity : 1 Vial
Brand Name : As per manufacturer
Cefoperazone And Sulbactam For Injection
Minimum Order Quantity : 10 Pieces
Dosage : As Directed by Physician
Indication : Treatment of various infections including respiratory, urinary tract, skin, intraabdominal, and gynecological infections.
Pacakaging (Quantity Per Box) : 1 Vial per Box
Quantity : 1.5 gm
Brand Name : As per manufacturer

 Send Inquiry
Send Inquiry
 Send Inquiry
Send Inquiry Send SMS
Send SMS
